Zembrin® is the world’s leading researched and clinically studied extract of Sceletium developed over more than a decade by an international team of scientists and physicians.
Zembrin® is an evidence-based botanical supplement that has been thoroughly studied for safety and efficacy in comprehensive preclinical as well as clinical studies.
Zembrin® is experiential – it has mood-enhancing, anxiety-reducing and cognitive function enhancing activities
Key Milestones in Our Story


Ancient Use by Khoi-San Communities


1st Patented & Standardized Sceletium Extract


8+ Published Clinical Studies


Clinically effective at a low dose (25 mg)


Works Within 2 Hours


Sustainable Cultivation & San Council Benefit Sharing


Global Recognition (Partners & Awards)
The Essence of Zembrin®
Four guiding pillars define who we are — our purpose, our promise, our principles, and our presence. Together, they express how Zembrin® blends science, ethics, and nature to enhance wellbeing worldwide.
Our Purpose
Elevating Everyday Wellness.
Zembrin® isn’t just a supplement; it’s a bridge between ancient wisdom and modern science.
Our Commitment
Where science meets nature.
At Zembrin®,
we don’t just promise results; we deliver them with integrity.
Our Ethos
Sustainability in every step.
From seed to shelf, Zembrin® is committed to sustainability and social responsibility.
Our Identity
More than a brand
Our presence is felt globally, from clinical studies to consumer products, all united by a common thread: enhancing human potential.
Science Meets Experience
“Backed by neuroscience. Rooted in tradition. Proven in real life.”
DAILY
BENEFITS
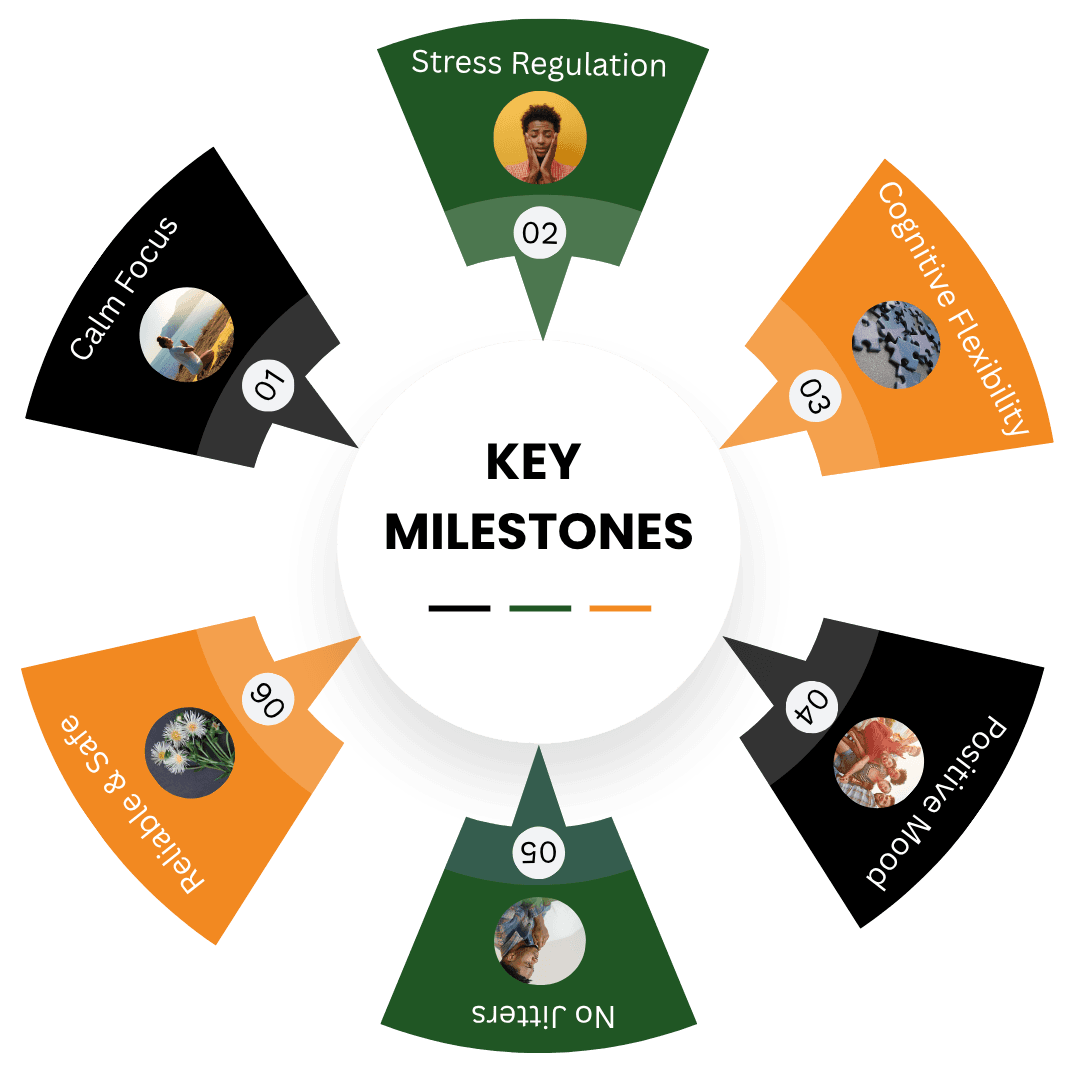

Calm Focus
Centered attention for deep work
Zembrin® supports balanced mental clarity, helping you stay present and focused without overstimulation — ideal for sustained concentration, creativity, and flow in work or study.

Stress Regulation
Lower perceived stress during busy blocks
Through its unique dual action on Serotonin and phosphodiesterase pathways, Zembrin® helps regulate stress responses, promoting composure and resilience even in demanding situations.

Cognitive Flexibility
Easier task-switching and problem-solving
Clinical studies show improved cognitive flexibility, allowing for quicker transitions between ideas and smoother adaptation to new challenges.

Positive Mood
Composed, uplifted outlook
Zembrin® gently enhances emotional balance and optimism, fostering a calm, uplifted state of mind that supports wellbeing throughout the day.

No Jitters
Calm energy, non-stimulating
Unlike caffeine or synthetic nootropics, Zembrin® delivers alert serenity — mental energy and clarity without spikes, crashes, or anxiety.

Reliable & Safe
Consistent, low-dose efficacy (25 mg)
Proven effective at a clinically validated 25 mg dose, Zembrin® provides predictable, sustained results with an excellent safety profile backed by multiple published studies.
🔬 The Science Behind Calm Focus
“Where neuroscience meets nature — bridging plant intelligence and human wellbeing.”


🌱 From Soil
“Cultivated through sustainable, traceable farming.”
🤝 With Purpose
“Royalties paid to the San Council — honouring heritage and collaboration.”


🔬 Through Science
“Extracted with care, backed by science, proven in wellness.”